Understanding the ocean’s changing chemistry
by Sarah Fesenmyer (NOAA Research Communications)
Ocean chemistry is changing faster right now than at any time over the past 50 million years. “We are fundamentally altering marine ecosystems,” says NOAA oceanographer Simone Alin, Ph.D., “by conducting the largest experiment imaginable.”
The experiment is the global release of carbon dioxide from fossil fuels into the atmosphere, now at a rate of around 9.5 billion metric tons of carbon per year. The oceans have absorbed approximately one-third of this additional carbon dioxide, which dissolves in seawater as carbonic acid and increases the acidity of seawater. With her colleagues at NOAA’s Pacific Marine Environmental Laboratory (PMEL), Alin is responsible for monitoring the rapidly changing chemistry of seawater and understanding the ramifications for the world’s oceans, particularly the highly productive, fisheries-rich coastal waters off the west coast of North America.
Earlier in her career, Alin studied historic impacts of humans on the environment, working at Lake Tanganyika in Africa, the second largest freshwater lake in the world. She used tiny fossils trapped in lake sediments to show a drop in biodiversity over the past few hundred years as people farmed ever steeper slopes, delivering an influx of sediment to the lake. Alin documented a profound and long-term impact on the lake ecosystem–but the human impact on the environment that she is currently researching is much bigger.
Ocean acidification is a global problem. The average pH of seawater has already decreased by 0.1, and is expected to decrease by another 0.3 units by 2100. Acidified waters affect marine life from corals to oysters to crabs by making it harder for some organisms to build skeletons or shells from calcium carbonate minerals dissolved in seawater, and may cause other impacts such as intensifying harmful algal blooms. Alin says that scientists are continually learning more about which life stages of which marine organisms are most vulnerable to increased acidity, whether because it affects their ability to form shells or causes physiological stress.
“Seawater pH and carbonate chemistry are changing so rapidly right now, compared to any time in geologic history that we know of,” she says. “We need high quality carbonate measurements across a range of coastal and offshore marine ecosystems to really understand the trend.”
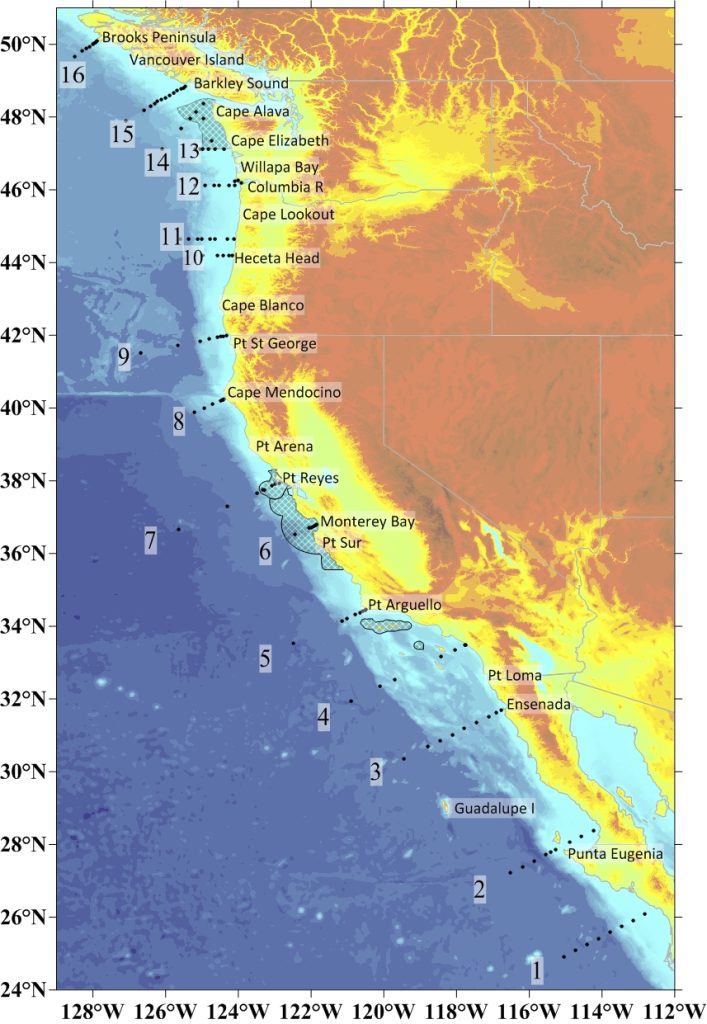
In May, Alin will be the Chief Scientist on the NOAA ship Ronald H. Brown during a monthlong research cruise to collect ocean acidification data on 16 transects of coastal waters from Canada to Mexico. She will coordinate between the captain of the 274-foot research vessel, the largest in NOAA’s fleet, and 35 scientists from government and university laboratories, collecting ocean data ranging from carbonate chemistry and temperature to samples of shell-forming plankton to indicators of harmful algal blooms. The cruise will help validate other ocean acidification data collection systems such as autonomous buoys, and provide a more complete picture of ocean conditions all along the West Coast.
Ocean acidification is affecting West Coast marine ecosystems sooner than many other parts of the world, in part because of the upwelling of deep ocean currents that are already naturally enriched with carbon dioxide. Alin joined her PMEL colleagues Nina Bednarsek, Ph.D., and Richard Feely, Ph.D., in a study where they showed that West Coast waters in some areas are now corrosive and undersaturated in carbonate minerals to the extent that pteropod shells are dissolving. Pteropods are tiny floating sea snails that are an important food source for salmon and other West Coast fisheries species, and their condition is an indicator of large changes developing in the ocean.
Planning the logistics of such a big research cruise has been intense, involving the efforts of many scientists from PMEL and partner institutions. Alin’s team is working out the water budget for the “rosette,” the sensor package that they will lower down to the seafloor during transects to measure temperature, salinity, and oxygen, and collect water samples for carbonate chemistry and other factors. The rosette is ringed with a series of 11-liter bottles. The scientists drop the rosette to the seafloor and have then programmed the instrument to fill and cap each bottle at specific depths on the way back up. Alin is making sure she allocates each scientist sufficient water samples at the specific ocean depths that they need for their research.
Because the Ronald. H. Brown ship has state-of-the-art laboratories, the researchers will be analyzing many of the samples they collect while onboard. When Alin studied carbon cycling in large tropical rivers as a postdoctoral fellow, she collected water samples by canoe from the Mekong and Amazon Rivers. She would then have to wait days to months to analyze her samples back in the laboratory. “This cruise will be amazing,” says Alin. “We will be running water samples 24/7, seeing the data immediately.”
She and her colleagues are particularly interested in seeing the condition of the Pacific coastal waters this spring, following a strong El Niño season, which affects ocean currents and upwelling, and the Pacific Ocean “Blob,” a large mass of warm water that was located off the West Coast for the last two years. Alin wants to know what effects these phenomena have had on pH, and also on phytoplankton, pteropods, and other zooplankton–the anchors of the marine food web.
Both PMEL and NOAA’s Northwest Fisheries Science Center will use net samples from the cruise to compare the abundance and condition of pteropods to pH and carbonate chemistry. “Very few research cruises sample from Baja to Canada in a one-month timeframe,” says Jennifer Fisher, a NOAA Fisheries researcher. “So it will be really helpful to get a dataset this large on the distribution of marine species to compare to changing ocean conditions.”
Alin became a government scientist so that she could tackle research that informs our country’s policy and management responses to complex environmental issues. Are there any practical responses for combatting ocean acidification? The West Coast Ocean Acidification and Hypoxia Science Panel recently outlined two possible approaches: using marine vegetation to capture carbon dioxide and convert it to plant tissue, or abiotic methods such as adding base chemicals to seawater to decrease its acidity. Alin is participating in a new research project to assess the effect of growing kelp in Puget Sound on seawater pH, with the hope that kelp will absorb excess carbon dioxide. She says that local measures such as this are most likely to work in partially-enclosed coastal water bodies.
“We need local measures to offset ocean acidification to buy ourselves time,” says Alin. “But the real problem that the world needs to solve is how to reduce global carbon emissions.”